Theoretical analysis of explosion behavior for CO/O2/N2, CO/O2/N2/H2O and CO/O2/N2/CO2 mixtures
Teoretická analýza výbuchových charakteristik směsí CO/O2/N2, CO/O2/N2/H2O a CO/O2/N2/CO2
Jan SKŘÍNSKÝ1, Ján VEREŠ1, Jana TRÁVNÍČKOVÁ1, Andrea DALECKÁ1
1Energy Research Centre, VSB-Technical University of Ostrava, corresponding author: jan.skrinsky@vsb.cz
Abstract
Gas mixture explosions and fires are responsible for most of the largest property loss events worldwide in the chemical and power industry. In this contribution, a theoretical analysis was performed of explosion behavior for CO/O2/N2, CO/O2/N2/H2O and CO/O2/N2/CO2 mixtures. Presented explosions based on real scenarios of accidents associated with transport and storage facilities with flammable chemicals. While explosions of pure flammable chemicals are well described in the literature, the information about explosions of toxic flammable substances is rather scarce. This work aims at studying the explosion behavior of pure mixture and of the inerted carbon monoxide-air mixtures at different initial temperatures and pressures. The results of mathematical modeling of the calculated maximum explosion pressure are presented.
Keywords: Gas explosion; Explosion modeling; CO
Abstrakt
Výbuchy a požáry hořlavých plynů v chemickém a energetickém průmyslu způsobují většinu z největších majetkových ztrát po celém světě. V tomto příspěvku je prezentována teoretická analýza chování výbuchu pro směsi CO/O2/N2, CO/O2/N2/H2O a CO/O2/N2/CO2. Prezentovaná data k výbuchům jsou založena na základě reálných scénářů havárií spojených s dopravou a skladováním zařízení s hořlavými látkami. Zatímco výbuchy chemicky čistých hořlavých látek jsou v literatuře popsány velmi dobře, informací o výbuších toxických hořlavých směsí látek je poměrně málo. Tato práce je zaměřena na studium chování výbuchu čisté směsi a inertizované směsi CO/O2/N2 a vzduchu za různých počátečních teplot a tlaků. V příspěvku jsou vůbec poprvé prezentovány výsledky matematického modelování maximálního výbuchového tlaku tohoto plynu.
Klíčová slova: Výbuchy plynů; Modelování výbuchů; CO
1. Introduction
Oxidation of solid, liquid and gaseous fuels either in oxygen atmosphere is surely attractive in the worldwide challenge for zero emission technology and in chemical process intensification. However, this opportunity raises several issues not only in terms of costs, but also in terms of safety. Indeed, in the case of loss of control, explosion consequences can be dramatic. Safety is a primary concern when dealing with flammable gases mixtures and the study of explosion pressures is an important consideration in addressing this issue (Lees, 2012). This work aims at studying the explosion behavior of CO/O2/N2 at different initial temperature and pressure and with one of two inerts H2O (vapor) and CO2 (gas). Nowadays, experimental tests are carried out in a closed cylindrical vessel. Experiments could be, however, expensive and time consuming, especially at elevated conditions of temperature and pressure, at which many industrial processes occur (Pekalski et al., 2005). Therefore, approximate calculation methods are being sought with which approximate values of explosion pressure can be calculated in reasonable time.
2. Analysis
Due to the complexity of the involved physical phenomena and to the lack of an adequate amount ofreliable experimental data, a number of different models and calculation procedures for estimating the physical consequences following the physical explosion of a gaseous state are presently reported in the literature. Due to the consistency of the input phys.-chem. data used in this study were chosen those from standard database (DIPPR, 2012) based mostly on experimental values with standard errors according to AIChE. The recommended values and temperature correlations are based on a critical, comprehensive evaluation by experts of the available data including inter-property relationships, inter-chemical relationships, and predicted/expected values.
Element balanced equation:
![]() (1)
(1)
where aj are moles of j-th element (atom); niare moles of i-th species; Ai,j number of j-th element in i-th species; l is the number of elements; j is the number of species.
The free energy (Helmholtz function) of the system F equation:
![]() (2)
(2)
where F is the free energy (Helmholtz function) of the system; ni are moles of i-th species; fi is free energy of i-th species.
![]() (3)
(3)
where fi is free energy of i-th species; λi are the Lagrangian multipliers for each element (j=1 to J); Ai,j number of j-th element in i-th species.
![]() (4)
(4)
where gi is the Gibbs free energy for single i-th species; ΔHfis the enthalpy of formation of the i-th species at standard conditions;
![]() (5)
(5)
where giis the Gibbs free energy for single i-th species; R is the universal gas constant; T is temperature; ni is the number of moles of every species; V is the volume.
![]() (6)
(6)
where aj are moles of j-th element (atom); niare moles of i-th species; Ai,j number of j-th element in i-th species; l is the number of elements; j is the number of species; gi is the Gibbs free energy for single i-th species; λi are the Lagrangian multipliers for each element (j=1 to J); R is the universal gas constant; T is temperature; ni is the number of moles of every species; V is the volume.
![]() (7)
(7)
where gi is the Gibbs free energy for single i-th species; Ai,j number of j-th element in i-th species; l is the number of elements; λi are the Lagrangian multipliers for each element (j=1 to J); R is the universal gas constant; T is temperature; ni is the number of moles of every species; V is the volume.
It has been shown (Skřínský et al., 2015) that the model is able to predict, with a reasonable accuracy in different fuel-enriched conditions, for different types of gaseous mixtures explosions. The equations 1-7 were used to investigate and quantify the role of initial temperature and pressure in affecting the explosion behavior of CO/O2/N2, CO/O2/N2/H2O and CO/O2/N2/CO2 mixtures.
3. Results and discussions
| Characteristic | Abbreviation | Unit | CO |
|---|---|---|---|
| Maximum explosion pressure | Pmax | bar(a) | 8.2G |
| 8.3CO | |||
| 8.0D |
Table 1: Explosion characteristics of flammable gas component
G = GESTIS-Substance database (IFA); CO = Compendium of Environmental Standards (CES); D = Design Institute for Physical Properties (AiChE).
Table 1 compares the theoretically derived data for the maximum explosion pressure of the studied gas mixture components. The values were adopted from the databases as the IFA and the AiChE, and from the literature as the Compendium of Environmental Standards (CES). In Table 1, for example the values of the Pmax adopted from IFA, AiChE and CEI (8.2 bar(a), 8.3 bar(a) and 8.0 bar(a), respectively) clearly appear to be quite different. All values were obtained by using standard testing conditions.
A) Adiabatic temperatures and explosion pressures for CO/O2/N2
Calculated adiabatic temperatures, Tad, and explosion pressures, Pex, for CO/O2/N2 were determined at various initial temperatures and ambient initial pressure. The results of calculations are given in Table 2 and Figures 1a-2a.
|
Fuel fraction [vol.%] |
Initial temperature (K) |
|||||||
|---|---|---|---|---|---|---|---|---|
| 298 | 358 | 418 | 478 | |||||
| Pex | Tad | Pex | Tad | Pex | Tad | Pex | Tad | |
| 15.0 | 5.94 | 1912 | 5.05 | 1953 | 4.41 | 1993 | 3.94 | 2033 |
| 30.0 | 7.98 | 2705 | 6.67 | 2710 | 5.74 | 2717 | 5.05 | 2724 |
| 45.0 | 7.78 | 2618 | 6.55 | 2647 | 5.68 | 2675 | 5.02 | 2701 |
| 60.0 | 6.43 | 2090 | 5.46 | 2133 | 4.77 | 2175 | 4.25 | 2218 |
| 75.0 | 4.76 | 1498 | 4.08 | 1542 | 3.60 | 1588 | 3.24 | 1633 |
Table 2: Computed explosion pressures for CO/O2/N2 mixtures at p0 = 1 bar(a)
From the numerical results presented in Table 2 it is possible to identify that the increase in the initial temperature lowers the maximum explosion pressure (37% for 30.0 vol. %). The value of the explosion pressure with varying CO/O2/N2 concentration is similar at all investigated initial temperatures. The maximum value of the explosion pressure is found close to 35 vol. % of CO for all fuel fraction vol. % (see Figure 1a). Further, in Table 3, we reported simulations on the explosion properties of CO/O2/N2 mixtures at various initial pressure and ambient initial temperature.
|
Fuel fraction [vol.%] |
Initial pressure (bar(a)) |
|||||||
|---|---|---|---|---|---|---|---|---|
| 1 | 5 | 10 | 15 | |||||
| Pex | Tad | Pex | Tad | Pex | Tad | Pex | Tad | |
| 15.0 | 5.94 | 1912 | 29.69 | 1913 | 59.38 | 1913 | 89.07 | 1913 |
| 30.0 | 7.98 | 2705 | 41.07 | 2897 | 83.08 | 2842 | 125.40 | 2864 |
| 45.0 | 7.78 | 2618 | 39.10 | 2633 | 78.28 | 2636 | 117.46 | 2638 |
| 60.0 | 6.43 | 2090 | 32.13 | 2090 | 64.26 | 2090 | 96.39 | 2090 |
| 75.0 | 4.76 | 1498 | 23.81 | 1498 | 47.62 | 1498 | 71.42 | 1498 |
Table 3: Computed explosion pressures for CO/O2/N2 mixtures at T0 = 298 K
From the data performed in Table 3 and Figure1b-2b, it is clear that the increase of initial pressure has significant effect on explosion pressure when only the air as oxidant.
a)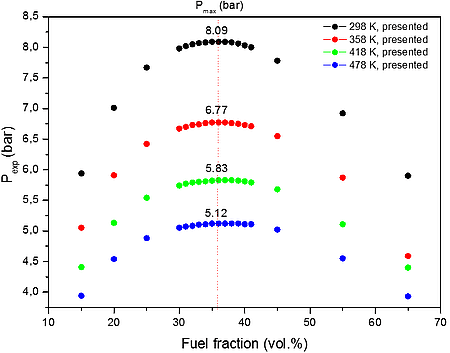
b)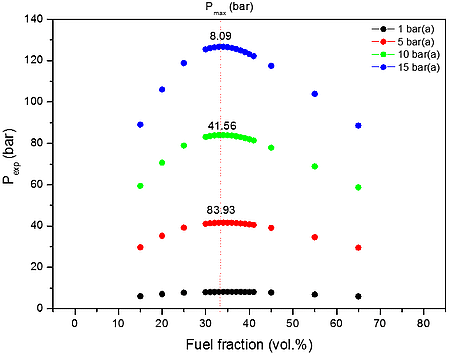
Figure 1: Calculated explosion pressure vs fuel fraction for explosions of a) CO/O2/N2 mixture at 298 K (top), 358 K (upper middle), 418 K (lower middle), and 478 K (bottom); b) CO/O2/N2 mixture at 1 bar(a) (top), 5 bar(a) (upper middle), 10 bar(a) (lower middle), and 15 bar(a) (bottom)
a)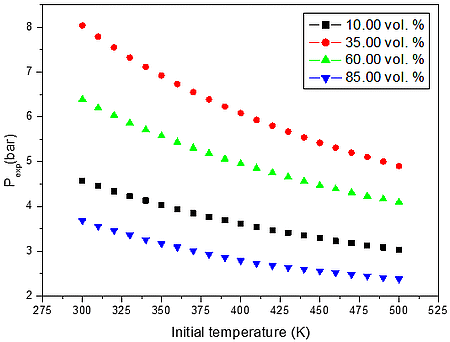
b)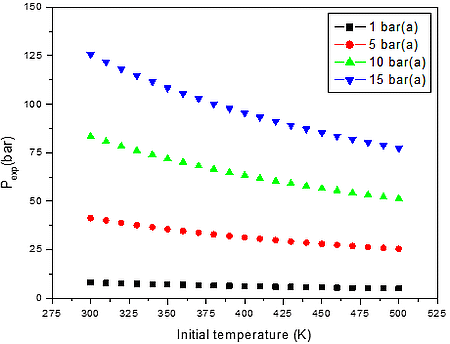
Figure 2: Calculated explosion pressure vs initial temperature for explosions of a) CO/O2/N2 mixture at 10.00 vol. % (top), 35.00 vol. % (upper middle), 60.00 vol. % (lower middle), and 85.00 vol. % (bottom); b) CO/O2/N2 mixture at 1 bar(a) (top), 5 bar(a) (upper middle), 10 bar(a) (lower middle), and 15 bar(a) (bottom)
B) CO2 and H2O inerting CO/O2/N2 mixtures
Further, maximum explosion pressure of CO/O2/N2/H2O and CO/O2/N2/CO2 at 300 K for four different fractions of H2O and CO2 was investigated. The initial ambient temperature profile estimation with different O2/N2 atmospheres similar to industrial application conditions was also analyzed. The numerical results of calculation are summarized in Figure 3. The flammability data for CO/O2/N2/H2O and CO/O2/N2/CO2 mixtures obtained in the present work provides adequate information for plant design purposes for a range of presented gas atmospheres, ambient temperature and pressures up to 15 bar(a).
a)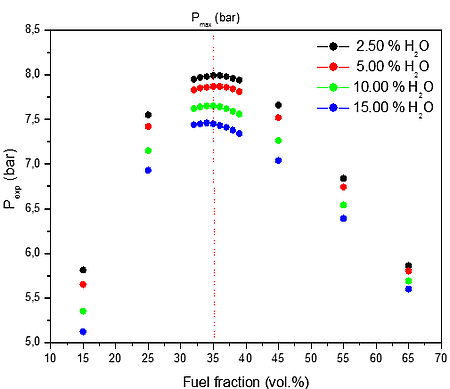
b)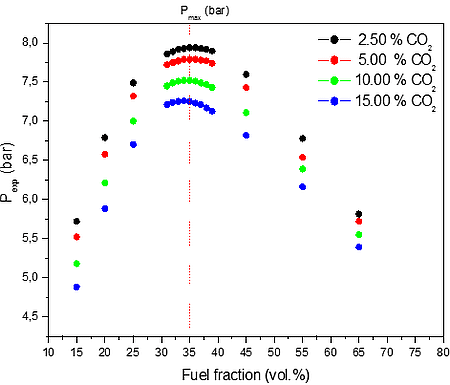
Figure 3: Calculated explosion pressure vs fuel fraction for explosions of CO/O2/N2 mixture at a) different initial H2O volume fractions 2.50 vol. % (top), 5.00 vol. % (upper middle), 10.00 vol. % (lower middle), and 15.00 vol. % (bottom); b) different initial CO2 volume fractions 2.50 vol. % (top), 5.00 vol. % (upper middle), 10.00 vol. % (lower middle), and 15.00 vol. % (bottom)
Figure 3 shows the calculated explosion pressure behavior of stoichiometric CO/O2/N2/H2O and CO/O2/N2/CO2 mixtures. The pressure trends for both mixtures are similar. Depending on the contents of H2O / CO2 and fuel fraction, they exhibit a single pressure peak at the maximum pressure. From Figure 3 it appears that the mixture reactivity, as quantified through Pmax, increases on decreasing the initial H2O / CO2 vol. % fraction. Furthermore, it can be noted that the explosion pressure is higher for the mixture with H2O for the same vol. % fraction values of fuel, in all cases. Therefore, based on equilibrium calculation presented in Figure 3 CO2 seems to be a better inert for CO/O2/N2 mixture. The results will be experimentally proofed in 1 m3 explosion apparatus.
4. Conclusion
The adiabatic explosion pressures and constant volume adiabatic explosion temperatures of CO2/O2/N2, CO/O2/N2/H2O and CO/O2/N2/CO2 mixtures at various initial temperatures and pressures were calculated. The model predictions for the CO2/O2/N2/H2O and CO/O2/N2/CO2 mixtures are compared for four different initial H2O / CO2 vol. % fractions. Finally, the results of simulations are compared to clarify the role of CO2 and H2O. Although the results from the evaluation indicate that presented theoretical simulations can become a valuable tool for rough estimation, the modeling requires further improvements to be useful for consequence modeling and design of industrial facilities. Thus, at the first stage, the equilibrium calculations can be used as a rough calculation of a worst case scenario. At the same time, these values will be used as approximate initial values for explosion experiments carried out in heated 1 m3 explosion apparatus designed by OZM Research s.r.o. at Energy Research Centre, VŠB - Technical University of Ostrava. The experimental data on the flammability of CO2/O2/N2, CO/O2/N2/H2O and CO/O2/N2/CO2 mixtures can readily be applied to the hazards of handling solutions of carbon monoxide gas mixtures. These conclusions apply only to solutions in closed vessels, but similar calculations based on gas-gas and vapor-gas equilibrium data can be made for solutions in opened vessels. The results represents a continuation of numerous efforts by various research groups, where the key underlying problem has been the understanding of results obtained in laboratory tests for predicting the consequences of gas/vapor explosion scenarios in industry (Skřínský et al., 2015).
5. Acknowledgment
This work would not have been possible without the financial support of two grants: (1) Innovation for Efficiency and Environment, reg. no. ED0036/01/01 supported by Operation Program Research and development for Innovation and financed by the Ministry of Education, Youth and Sports; (2) Innovation for Efficiency and Environment - Growth, reg. no. LO1403 supported by National Programme for Sustainability and financed by the Ministry of Education, Youth and Sports. J.V. is thankful for the financial support of the project “Support research and development in the Moravian-Silesian Region 2014 DT 1-International research teams” 02722/2014/RRC, financed from the budget of the Moravian-Silesian Region.
6. Literature
[1] LEES, F. P. Loss Prevention in the Process Industries: Hazard Identification, Assessment, and Controls: volume 1-3. Oxford: Butterworth-Heinemann, 2012.
[2] PEKALSKI, A. …[et al.]. Determination of the Explosion Behaviour of Methane and Propene in Air or Oxygen at Standard and Elevated Conditions. Process Saf. Environ, 2005, vol. 83, s. 421-429.
[3] SKŘÍNSKÝ, Jan …[et al.]. Explosion chracteristics of methane at elevated initial temperatures. Časopis výzkumu a aplikací v profesionální bezpečnosti [online], 2015, roč. 8, č. 2-3. Dostupný z WWW: <http://www.bozpinfo.cz/josra/josra-02-03-2015/methane-explosion-characterist.html>. ISSN 1803-3687.
[4] GESTIS-Substance database. Institut für Arbeitsschutz der Deutschen Gesetzlichen Unfallversicherung (IFA), 2015.
[5] Design Institute for Physical Properties. DIPPR Project 801: Full Version. Design Institute for Physical Property Research/AIChE, 2012.
Vzorová citace
SKŘÍNSKÝ, J. …[et al.]. Theoretical analysis of explosion behavior for CO/O2/N2, CO/O2/N2/H2O and CO/O2/N2/CO2 mixtures. Časopis výzkumu a aplikací v profesionální bezpečnosti [online], 2015, roč. 8, č. 4. Dostupný z WWW: <http://www.bozpinfo.cz/josra/josra-04-2015/analysis-explosion-behavior.html>. ISSN 1803-3687.
Užitečné odkazy
Provozovatel portálu
Jeruzalémská 1283/9
110 00 Praha 1


